********************
25.-28. June 2017 Joint Meeting on Medicinal Chemistry, Dubrovnik, Croatia
Sheraton hotel Srebreno (6 km far from centre of Dubrovnik and 12 km far from airport).
hotel Sheraton Dubrovnik Riviera Hotel inSrebreno at the Dubrovnik Riviera (www.sheratondubrovnikriviera.com )
Conference web page: http://www.jmmc2017.hr/
vesna.gabelica.markovic@hkd.hr
Registration fee 400 E (Academia) 150E (student) includes:
Participation in all scientific sessions
Entrance to the exhibition area
Congress bag with congress material
Coffee breaks
Lunches
Participation in the Opening Ceremony and Welcome Reception
Guided tour through Dubrovnik centre (proposal)
----------------------------
1. Country plenary lecturers: No conference fee. We, local organizing committee, cover accommodation.
2. Key note lecturers: Conference fee will be the same as students
3. One representative from each country has no fee and we cover accommodation.
--------------------
Names of plenary and keynote lectures June/July 2016
Short CVs of Plenary lecturers September/October 2016
Early Registration Fee Febrary 2017
Abstract Submission Deadline
kolegov zo SAV a pripadne doc Berkesa. Dr. Stefeka (Dr. Majekova, Dr. Soltesova-Prnova)
The preferred topics are
Anti-infectives; Neuroactive drugs; Anticancer drugs
Epigenetic modulation
New chemical entities from nature or inspired by nature;
Redox active compounds
Small molecule probes
Computer-aided drug design and discovery; Structure-activity relationships
Drug delivery and resistance; Combinatorial therapy
Emerging strategies in drug discovery and drug development
Plenary lectures/Key note lectures/Speakers
Plenary lecture (40 min):
Prof. Andej Boháč (anticancer kinase inhibitors) Comenius University, Slovakia
Key note lecture (30 min):
Richard Engh (24.05.2016 projekt, Norway) -----------24.05.2016
"Addressing dynamic structural elements of emerging protein kinase drug targets in oncology and neurodegenerative disease with 'special interactions': sulfur, halogens, and arene moieties"
Associated prof. Andrej Boháč, PhD. is a medicinal, bioorganic, organic chemist, researcher and lecturer at the Comenius University in Bratislava, Slovakia [https://fns.uniba.sk/]. He is also a CEO of SME research company Biomagi, Ltd. [http://www.orsr.sk/hladaj_subjekt.asp?OBMENO=biomagi&PF=0&SID=0&R=on&lan=sk and https://mch.estranky.sk/clanky/biomagi.html]. The company was composed by him in 2010.
A. Boháč was or is a member of several chemistry / biology COST networks [http://www.cost.eu/about_cost/who/%28type%29/5/%28wid%29/15592, CM0602, CM1106 and CA1535]. Within the COST he developed several international collaborations (e.g.: France – Strasbourg, USA – New Jersey, Spain – Malaga, Turkey – Ancara, Italy – Siena, Slovenia – Ljubljana, Switzerland - Bellinzona).
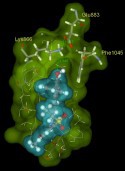
A. Boháč research interest is involved in conformational kinase, ligand / target interaction analyses and in SBDD (Structure Based Drug Design) what means development (computer predictions / organic synthesis / bioactivity evaluation / optimization) of Clk, Axl, Mer, VEGFR2 kinase inhibitors (potential oncology therapeutics) and modulator of SMO GPCR (target is a part of the Hedgehog pathway involved in stem cells biology useful for development of anticancer or regenerative medicine drugs). Within his research he identified an unknown conformation of tyrosine kinases possessing special DFG-in/out arrangement with an opened A-loop [1] Lintnerova L. et al. 2014 EJMECH + unpublished results (ligand supported assembly and stabilisation of unusual kinase conformation). A. Boháč and his colleagues developed several powerful VEGFR2 TK inhibitors [1-3] Lintnerova L. et al. and Vojtickova M. et al. EJMECH 2014-2015, Remko M. et al. Structural Chem 2011. Recently they developed powerful CLK-1 inhibitors based on N-aryloxazol-2-amine and chromeno[2,3-b]pyridin-2-one skeletons all of them possessing sulphur and aryl moieties. The biological activities of the above compounds were determined by radiometric enzymatic assay by ProQinase in Germany: IC50 (CLK1): 20, 20, 30, 40 and 80 nM. [A. Bohac et al. 2016, unpublished results]. The binding poses in CLK-1 kinase for four of the above inhibitors were predicted. These strong CLK1 inhibitors can help establish crystallization and can be a part of structure-binding strength studies. What more, to have an X-ray from the above ligand / kinase complexes enable us to design the second generation of inhibitors more precisely and also open us the possibility to perform design for new up to day unknown inhibitors (e.g. Clk1-3 DFG-out inactive conformation or any of Clk4 and Axl TK structures are not yet known in the literature).
A. Bohac also developed an effective prediction methodology to select target active structures from up to 14 millions of commercially available compounds. Recently by this methodology he identified eight novel TAM TK inhibitors IC50: 3, 8, 10, 24, 51, 58, 643, 985 uM (MER TK) and by cooperation with prof. Raymond B. Birge (Rutgers, New Jersey Medical School University Hospital Cancer Center, USA) also their Tyro3 AXL and Mer cell activity profiles were determined on chimeric EGFR-TAM CHO cell lines. A. Bohac and his co-workers predicted and obtained 11 novel compounds that are just ready to be screened on Axl TK activities.
[1] L. Lintnerová, M. García-Caballero, F. Gregáň, M. Melicherčík, A. R. Quesada, J. Dobiaš, J. Lác, M. Sališová, A. Boháč; A development of chimeric VEGFR2 TK inhibitor based on two ligand conformers from PDB: 1Y6A complex - Medicinal chemistry consequences of a TKs analysis 2014 EJMECH 72 146-159, DOI:10.1016/j.ejmech.2013.11.023.
[2] M. Vojtičková, J. Dobiaš, G. Hanquet, G. Addová, R. Cetin-Atalay, D.C. Yildirim, A. Boháč, Ynamide Click chemistry in development of triazole VEGFR2 TK modulators, 2015 EJMECH 103 105-122, doi:10.1016/j.ejmech.2015.08.012.
[3] M. Remko, A. Boháč, L. Kováčiková; Molecular structure, pKa, lipophilicity, solubility, absorption, polar surface area, and blood brain barrier penetration of some antiangiogenic agents, 2011 Structural Chemistry 22 2011 635-648.