BIOMAGI

TARIC Code 2942000000 (other organic compounds) Only for laboratory testings, not for use in humans or animals, only smallest 5 mg amounts
Legal Name: Biomagi, Ltd.
PIC number: 936 254 477
History: Biomagi, Ltd. was composed 23rd March 2010 by prof. RNDr. Andrej Boháč, PhD. who became its CEO.
Chief Executive Officer: prof. RNDr. Andrej Boháč, PhD. (CV-17. August 2016)
Legal Address: Mamateyova 26/1508, 851 04 Bratislava, Slovakia
Bank Address: ČSOB bank, Námestie hraničiarov 33, 851 03 Bratislava, Slovakia
Bank Account IBAN: SK91 7500 0000 0040 2602 0171
SWIFT: CEKOSKBX
Business Registration Number: 45436819
VAT Number: SK2022999759
EORI Number: SK2022999759
Registration web page: http://www.orsr.sk/hladaj_subjekt.asp?OBMENO=biomagi&PF=0&SID=0&R=on&lan=en
Web page: https://mch.estranky.sk/clanky/biomagi.html
Phone: +421 902 522 144
E-mail: andrej.bohac@uniba.sk or biomagibiomagi@gmail.com
Topics: Medicinal chemistry, preclinical drug candidates development, structure based drug design, virtual high throughput screening, drug like filters, target selection and analysis, ligand / protein intermolecular interaction analysis, molecular modelling and lead optimisations, cancer drug candidates and cancer stem cells modulators development (inhibitors of tyrosine kinases VEGFR2, CLK1, TAM TKs, HDAC2, SMO in Hedgehog pathway, PFKFB3 and ALR2 against diabetes late complications), advanced organic synthesis and full spectral compound characterisation (NMR, IR, UV, MS).
------------------------------------------19.02.2021
Biomagi Ltd. (BM) is a creative, medicinal chemistry focused SME, composed in 2010. The BM is specialized on a rationale and fast development of novel drug candidates for broad scope of biological targets. BM collected (and is continuously updating) the database consisting of 8.7 millions (January 2021) diverse organic structures of commercially available compounds. This unique database can be used to target a required bio-macromolecules of desired pathogen based on their known 3D structure. The BM is specialized on fast sending of identified, precisely selected and validated drug-like compounds directly for biological screening. At this point (time and resources consuming) synthesis is not needed for identification of novel biologically active compounds that use to have a high potential to modulate an activity of the selected biological target. Within 2 months after finalization of Biomagi computer predictions, the structure and purity validated compounds will be sent to a partner for screening. The Biomagi usually requires getting back results from assays to creatively continue further development towards drug candidates (structure optimization, development of derivatives, novel analogues, bioisosteres etc). From July 2020 we have predicted and already supported 49 inhibitors focused against SARS-CoV-2 activity.
Best wishes, Andrej Bohac (CEO and owner of the Biomagi)
also at Comenius University in Bratislava
Faculty of Natural Sciences
Ilkovicova 6, Mlynska dolina
842 15 Bratislava
Slovakia
------------------Bimoagi, Ltd. (BM)
Biomagi, s.r.o. vie zabezpecit pocitacom predpovedane vzorky
v publikaciach, ako aj na moznom patente, ako 10 % spoluvlastnika)
--------------------
Biomagi Ltd. (BM) is a medicinal chemistry research SME, composed in 2010. The BM is specialized on a rationale and fast development of novel small organic drug candidates for a broad scope of biological targets. Recently, BM collected (and is continuously updating) the database consisting of 8.5 millions of diverse organic structures of commercially available compounds. The Biomagi database can be used to target a required bio-macromolecule that has its structure already known. The BM is specialized on sending of identified, precisely selected and validated drug-like compounds to a partner for biological screening. The big advantage is, that at this point (time and resources consuming) organic synthesis is not needed for development of novel leading compounds. The supported compounds (antagonist or agonist candidates) use to have a high potential to modulate an activity of the selected biological target. Within 1.5-2 months after finalization of Biomagi predictions, the structure and purity validated compounds will be sent to a partner for screening. The Biomagi requires get back results from assays in order to creatively continue on further development towards drug candidates (structure optimization, development of derivatives, novel analogues, bioisosteres etc). In a very recent time we have analyzed all available PFKFB3 conformers in PDB, preformed predictions and now obtained 7 novel PFKFB3 inhibitor candidates for our research purposes.
Best wishes, assoc. prof. RNDr. Andrej Bohac, PhD. (CEO and owner of the Biomagi)

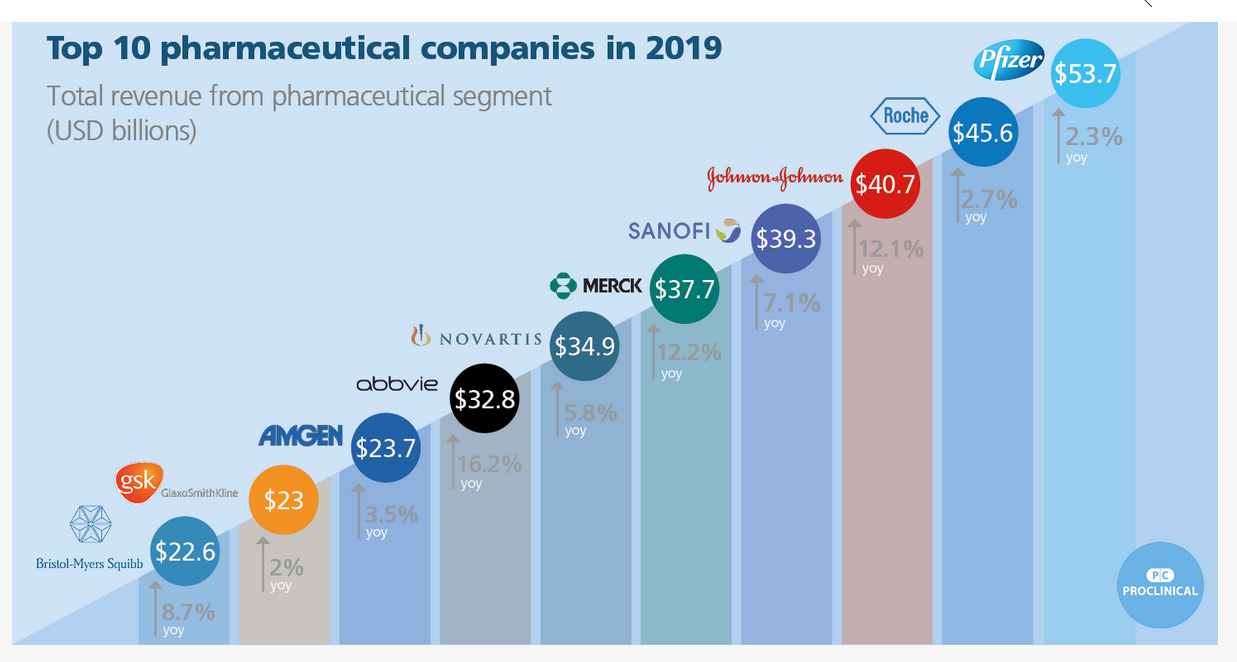
* https://www.proclinical.com/blogs/2019-3/the-top-10-pharmaceutical-companies-in-the-world-2019
No. of full time employees: 1
No. of full time employees in R&D: 1
Annual turnover approx.: 25 000 Euro
Infrastructure: complementary with state-of-the-art for multidisciplinary medicinal chemistry research. There are two laboratories for molecular modelling, equipped with four local workstations and with access to supercomputer centers including large (>1000 CPU) cluster. There is one biological laboratory as well as three laboratories equipped for organic synthesis, including access to high field NMR (300 and 600 MHz), FT UV and IR equipments, high-resolution mass spectroscopy, as well as facility for parallel synthesis and microwave or ultrasound reactions.
specializacie pod odborom chemia
1. medicinska chemia [1]
2. chemo informatika [2]
3. chemicka biologia (bioortogonalne reakcie) [3]
4. organicka chemia [3]
5. chemicka synteza [4]
6. bioorganicka chemia [5]
7. stereochemia [6]
8. enantioselektivna synteza (stereoselektivna synteza) [7]
9. spectroskopia (organicke zluceniny) [8]
[1] https://en.wikipedia.org/wiki/Medicinal_chemistry
[2] https://en.wikipedia.org/wiki/Cheminformatics
[3] https://en.wikipedia.org/wiki/Chemical_biology
[4] https://en.wikipedia.org/wiki/Organic_chemistry
[5] https://en.wikipedia.org/wiki/Chemical_synthesis
[6] https://en.wikipedia.org/wiki/Bioorganic_chemistry
[7] https://en.wikipedia.org/wiki/Stereochemistry
[8] https://en.wikipedia.org/wiki/Enantioselective_synthesis
[9] https://en.wikipedia.org/wiki/Spectroscopy
Skúsenosti využiteľné pre projekt Impulz Corona: v rámci (1) identifikácia kľúčového biologického cieľa, štruktúr a aktivít známych inhibítorov a liečiv (využitie databáz SciFinder, Reaxys a (2)), racionálny vývoj (predikcia, zaobstaranie, testovanie) kandidátov nových nosných štruktúr (inhibítory, alebo promótory biologických cieľov) pre nové antivirotiká (1) spolu s Biomagi DB 8.5 M dostupných zlúčenín a (3), syntéza nedostupných, alebo optimalizácia identifikovaných kandidátov na antivirotiká (4-9).
-------------------------------XFEL SK-----------------------------
Pre rýchly racionálny vývoji modulátorov biologických cieľov (inhibítor / promótor; antagonista / agonista) treba veľkú knižnicu variabilných štruktúr dostupných organických zlúčenín. Po virtuálnom testovaní a interakčnej analýze sa vybrané zlúčeniny dajú zaobstarať a použiť na testovanie, alebo na tvorbu komplexov s biomakromolekulami. Týmto prístupom sa veľmi urýchli vývoj ligandov lebo nie je treba predpovedané látky syntetizovať.
Preto sme vytvorili pravidelne aktualizovanú databázu 8.5 milióna štruktúr dostupných zlúčenín od svetových dodávateľov a iných zdrojov (posledná verzia BM-0420). Knižnicu sme filtrovali a ponechali len ligandy s vlastnosťami liečiv a ďalšími vhodnými fyzikálno-chemickými vlastnosťami (napr. FW ≤ 500; LogP ≤ 5; N,O ≤ 10; NH,OH ≤ 5; NRB ≤ 10, PSA ≤ 140 a iné napr. bez náboja, bez kovu, redukovaný počet Cl, Br, P, Si atómov...). Odstránené boli aj štruktúry obsahujúce reaktívne alebo toxické funkčné skupiny. Nakoniec sme odfiltrovali aj chirálne zlúčeniny (ca 30%) lebo sú komplikáciou pre ďalší vývoj liečiv. K finálnej licencii pre chirálne liečivá treba testovať obidva enantioméry, ako aj racemát. Podstatne náročnejšia je aj enantioselektívna syntéza takýchto zlúčenín. Samozrejme, chirálne zlúčeniny je tiež možne zapojiť do predikcii, ale na počiatku by som volil achirálne látky, ktorých je momentálne dostatok na úspešnú identifikáciu vhodných ligandov. Vyššie uvedeným spôsobom sme z 8.5 milióna štruktúr vyselektovali tie, ktoré majú vhodné farmakokinetické vlastnosti a predpoklady byť ligandom s dobrou afinitou k vybranému biologickému cieľu. Z takto filtrovanej knižnice 2D štruktúr sme generovali energeticky výhodné 3D štruktúry konformérov, tautomérov a protomérov pri fyziologickom pH krvi (pH = 7.3). Nakoniec sme získali 3.47 milióna štruktúr vhodných pre počítačové predikcie biologicky aktívnych zlúčenín určených pre testovanie a štruktúrnu analýzu ich komplexov. Od počiatku po získanie predpovedaných zlúčenín (filtrovanie knižnice, príprava ligandov, príprava 3D štruktúry biomakromolekuly, počítačové predikcie dokovaním, analýzy interakcií, selekcia ligandov, zaobstaranie zlúčenín, potvrdenie štruktúry a analýza čistoty HPLC MS, HRMS) treba počítať 2-3 mesiace a priame náklady 350-450 Euro + DPH / 4 mg predpovedanej zlúčeniny. Toto platí pri zaobstaraní min. 10 a viac ligandov pre vybraný biologický cieľ, ktorého 3D štruktúra je dostupná v PDB alebo interne. Výsledkom spolupráce by mali byť publikácie, patenty, prípadne participácia na ďalšom vývoji či licencovaní liečiv. Takýmto spôsobom sme vyvinuli ligandy pre VEGFR2 TK (tumor angiogensis, prized with Pfizer Research Award 2009), CLK-1, TAM TKs, HDAC2 (antitumor targets), PFKFB3 (metabolism - tumor glycolysis), SMO receptor in Hedgehog pathway (tumor stem cells), ALR2 (chronic diabetes complications) a iné. Teraz sa zameriavame na antivirálne ciele ako Mpro, PLpro, Spike (S1 RBD) / ACE2, Furin, Sigma R1, Cathepsin. Naša aktivita však týmito biologickými cieľmi nie je limitovaná.
-------------------------------XFEL EN-----------------------------
The fast, rational development of modulators (ligands: inhibitor / promoter; antagonist / agonist) for biological targets requires an access to a large library of variable structures of readily available organic compounds. After virtual screening and interaction analysis, selected compounds can be obtained and directly used for testing or to form complexes with biomacromolecules. This approach greatly accelerates the development of target active ligands because there is no need to synthesize the predicted compounds.
Therefore, we have created a regularly updated database of 8.5 million structures of available compounds from global suppliers and other sources (latest version BM-0420). We have also filtered the library and left only ligands with drug like and other suitable physicochemical properties (e.g. FW ≤ 500; LogP ≤ 5; N, O ≤ 10; NH, OH ≤ 5; NRB ≤ 10, PSA ≤ 140 and others e.g. without charge or metal atom, with reduced number of Cl, Br, P, Si atoms ...). Structures containing reactive or toxic functional groups were removed. Finally, we also filtered off all chiral compounds (ca. 30 %) as they are a complication in further drug development. Both the enantiomers as well as the racemate must be verified for the final market license of chiral drugs. Enantioselective synthesis of such compounds can be also much more difficult compare to other compounds. Of course, chiral compounds can also be involved in our predictions, but initially I would prefer achiral substances that represent currently satisfying amount to successfully identify suitable ligands. From 8.5 million structures and the above described method, we have selected those, that have suitable pharmacokinetic properties and predisposition to be ligands for the selected biological target. From such a filtered sub-library of 2D structures, energetically advantageous 3D structures of conformers, tautomers and protomers at physiological blood pH (pH = 7.3) were generated. Recently, by this method, we have obtained 3.47 million structures suitable for further computer-based predictions of biologically active compounds dedicated for direct testing and structural analysis of their complexes. We need 2-3 months from the beginning to obtaining the predicted compounds (library filtering, preparation of ligands, preparation of 3D structure of the biomacromolecule (target), docking, interaction analysis, ligand selection, compound obtaining, their structure confirmation and purity analysis by HPLC MS / HRMS) with direct costs 350-450 Euro + VAT / 4 mg for one predicted compound. This is valid when procuring min. 10 or more ligands for a selected target whose 3D structure is available in the PDB or internally. As results of the cooperation, publications, patents, or participation in the further development or licensing of drugs is expected. By the above method, we already developed several ligands for VEGFR2 TK (tumor angiogensis, prized with Pfizer Research Award 2009), CLK-1, TAM TKs, HDAC2 (all as antitumor targets), PFKFB3 (metabolism - tumor glycolysis), SMO receptor in the Hedgehog pathway (tumor stem cells ), ALR2 (chronic diabetes complications) and others. Now, we are focused on SARS CoV-2 targets such as Mpro, PLpro, Spike (S1 RBD) / ACE2, Furin, Sigma R1 and Cathepsin. However, our activity is not limited by these biological targets and we are open for further cooperation.
------------------------------------------------------------------------------------------------------------------
LIST OF PUBLICATIONS AND PATENTS:
ORCID Andrej Boháč: 0000-0002-5233-9466, SCOPUS Andrej Boháč: 6602628612
Publications: 2020 J Med Chem 63 369−381 (Reg EVP: 13.01.20) PDF, M. Hlavac, L. Kovacikova, M. Soltesova Prnova, P. Sramel, G. Addova, M. Majekova, G. Hanquet, A. Bohac, M. Stefek (DOI: 10.1021/acs.jmedchem.9b01747) Development of Novel Oxotriazinoindole Inhibitors of Aldose Reductase: Isosteric Sulfur/Oxygen Replacement in the Thioxotriazinoindole Cemtirestat Markedly Improved Inhibition Selectivity 20,10,5,12,12,6,5,20,10
2019 Gen Physiol Biophys 38 157-163 (Reg EVP: MZ-2019) PDF, J. Horváthová, R. Moravčík, A. Boháč, M. Zeman (DOI: 10.4149/gpb_2018047) Synergic effects of inhibition of glycolysis and multikinase receptor signalling on proliferation and migration of endothelial cells
2019 Chem Papers 73 71-84 (Reg EVP: 20.2.2019) PDF, ESM, J. Dobiaš, M. Ondruš, M. Hlaváč, M. Murár, J. Kóňa, G. Addová, A. Boháč (DOI: 10.1007/s11696-018-0576-6) Medicinal Chemistry - an effect of a desolvation penalty of an amide group in development of kinase inhibitors(35/5/5/5/5/10/35: 20%CHU)
2019 Chem Papers 73 55-69 (Reg EVP: 20.2.2019) PDF, ESM, M. Lácová, M. Čakurda, P. Koiš, G. Addová, A. Boháč (DOI: 10.1007/s11696-018-0566-8) A novel and efficient synthesis of 3-aryl-5-(2-hydroxybenzoyl)pyridin-2(1H)-ones by re-cyclization of N-(oxopyranochromenyl)acetamides and their antineoplastic screening(10/35/10/10/35, 20%CHU)
2018 Chem Papers 72 2979-2985 (Reg EVP: 3.10.18), (DOI: 10.1007/s11696-018-0548-x), PDF, ESM, M. Murár, J. Horvathová, R. Moravčík, G. Addová, M. Zeman, A. Boháč, Synthesis of glycolysis inhibitor (E)-3-(pyridin-3-yl)-1-(pyridin-4-yl)prop-2-en-1-one (3PO) and its inhibition of HUVEC proliferation alone or in a combination with the multi-kinase inhibitor sunitinib
2018 Chem Papers 72 683-690 PDF, ESM, M. Čakurda, P. Koiš, G. Addová, M. Lácova, A. Boháč (DOI: 10.1007/s11696-017-0319-0) 2H-Pyrano[3,2-c]quinolin-2-ones - their convenient synthesis and selected reactions
2017 ARKIVOC V, 204–215 PDF, ESM, I. Zemanová, R. Gašparová, A. Boháč, T. Maliar, F. Kraic, G. Addová; (DOI: https://doi.org/10.24820 /ark.5550190.p010.240) Synthesis and antibacterial activity of furo[3,2-b]pyrrole derivatives.
2017 Beilstein J Org Chem 13, 1350–1360 pdf + supp mat or links in BJOCH: PDF + ESM J. Dobiaš, M. Ondruš, G. Addová, A. Boháč; (DOI: 10.3762/bjoc.13.132) Switchable highly regioselective synthesis of 3,4-dihydroquinoxalin-2(1H)ones from o-phenylenediamines and aroylpyruvates
2017 ARKIVOC IV, 184–193 DOI: http://dx.doi.org/10.3998/ark.5550190.p009.788, PDF, I. Zemanová, R. Gašparová, F. Kraic, D. Kružlicová, T. Maliar, A. Boháč, G. Addová, Synthesis of furo[2',3':4,5]pyrrolo[1,2-d][1,2,4]triazine derivatives and their antibacterial activity.
2017 Eur J Med Chem 126, 754–761 link to the paper in EJMECH + PDF + suppl mat + videa (DOI: 10.1016/j.ejmech.2016.11.003) M. Murár, J. Dobiaš, P. Šramel, G. Addová, G. Hanquet, A. Boháč; Novel CLK1 Inhibitors Based on N-aryloxazol-2-amine Skeleton - a Possible Way to Dual VEGFR2 TK / CLK Ligands
2017 RCS MedChemComm 8, 81-87 (DOI: 10.1039/c6md00392c, pdf + supplementary info) Deniz Cansen Kahraman, Gilles Hanquet, Loïc Jeanmart, Steve Lanners, Peter Šramel, Andrej Boháč, Rengul Cetin-Atalay; Quinoides and VEGFR2 TKIs influence the fate of hepatocellular carcinoma and its cancer stem cells.
2016 Farmaceutický Obzor 85(5-6) 99-112 LINK CASOPIS, PDF CLANKU Základná terminológia vo farmaceutickej a medicínskej chémii, M. Remko, A. Boháč
2016 Cell Commun & Signaling 14:19 (DOI: 10.1186/s12964-016-0142-1, pdf) S.G. Kimani, S. Kumar, V. Davra, Y.-J. Chang, C. Kasikara, K. Geng, W.-I Tsou, S. Wang, M. Hoque, A. Boháč, A. Lewis-Antes, M.S. De Lorenzo, S.V. Kotenko, R.B. Birge; Normalization of TAM post-receptor signaling reveals a cell invasive signature for Axl tyrosine kinase.
2016 Gen Physiol Biophys 35 511-514 (DOI: 10.4149/gpb_2015055, pdf) R. Moravčík, K. Stebelová, A. Boháč, M. Zeman, Inhibition of VEGF mediated post receptor signalling pathways by recently developed tyrosine kinase inhibitor in comparison with sunitinib
2015 Eur J Med Chem 103 105-122. DOI:10.1016/j.ejmech.2015.08.012, pdf, suppl mat ) Margaréta Vojtičková, Juraj Dobiaš, Gilles Hanquet, Gabriela Addová, Rengul Cetin-Atalay, Deniz Cansen Yildirim, Andrej Boháč; Ynamide Click chemistry in development of triazole VEGFR2 TK modulators.
2015 J Het Chem 52 425-39, DOI: 10.1002/jhet.2063, pdf, Lucia Lintnerová, Lucia Kováčiková, Gilles Hanquet, Andrej Boháč, Selected Methodologies Convenient for the Synthesis of N,5-Diaryloxazole-2-amine Pharmacophore
2014 Eur J Med Chem 72, 146-159 DOI:10.1016/j.ejmech.2013.11.023, pdf + supplement mat (plachty a spektrá), Lucia Lintnerová, Melissa García-Caballero, Fridrich Gregáň, Milan Melicherčík, Ana R. Quesada, Juraj Dobiaš, Ján Lác, Marta Sališová, Andrej Boháč A development of chimeric VEGFR2 TK inhibitor based on two ligand conformers from PDB: 1Y6A complex - Medicinal chemistry consequences of a TKs analysis (SymAsym, klasifikácia všetkých 34 PDB VEGFR2 TK, objav netradičných DFG-out A-loop opened 1Y6A-like TK konformérov)
2013 Central Eur J Chem 11 502-513 Renata Gašparová, Pavol Koiš, Margita Lácová, Silvia Kováčová, Andrej Boháč Synthesis, reactions and antineoplastic activity of 3-(2-oxo-2H-chromen-3-yl)-2-oxo-2H,5Hpyrano[3,2-c]chromene derivatives
2013 Beilstein J Org Chem 9, 173-179 DOI:10.3762/bjoc.9.20, pdf, M. Murár, G. Addová, A. Boháč; Synthesis of 5-(ethylsulfonyl)-2-methoxyaniline: An important pharmacological fragment of VEGFR2 and other inhibitors.
Patents:
Lacova, M.; Bohac, A.; Biologically active compounds with pyrano[3,2-c]chromen-2(5H)-one skeleton and a method of their preparation Slovakia (2011), SK 287542 B6 20110104.
Bohac, A.; Snuparek, V.; Varga, I.; Gattnar, O.; Skoda, A.; Manduch, M. Method of production of trans-tramadol and its salts by epimerization of cis-tramadol Slovakia (2004), SK 284181 B6 20041005.
Veverka, M.; Boháč, A.; Almássy, A.; Addová, G.; Varga, I.; Method of preparation of derivatives of substituted 2-arylalkanoic acids by rearrangement of alpha-haloacetophenones and analogs in the presence of orthoformates and acid catalysts Patent No. WO 2004/046080 A1 – Geneva, World Intellectual Property Organization, 2004.
Veverka, M.; Boháč, A.; Križ, M.; Varga, I.; Method of preparation of terfenadine and its derivatives, Patent No. WO2004/043922 A1. Geneva, World Intellectual Property Organization, 2004.
----------------------------------------------------------------------------------------------------------- some older publications and patents
M. Ilić, J. Ilaš, P. Dunkel, P. Mátyus, A. Boháč, S. Liekens, D. Kikelj, Novel 1,4-benzoxazine and 1,4-benzodioxine inhibitors of angiogenesis, 2012 Eur. J. Med. Chem. 58 160-170.
B. Gabajová, D. Valková, A. Boháč, E. Kováčová, R. Moravčík, M. Zeman; hVEGF165 Expression in Escherichia coli Conserves Its Biological Function 2012 J. of Chem. and Chem. Engineering 6 738-743.
M. Remko, A. Boháč, L. Kováčiková; Molecular structure, pKa, lipophilicity, solubility, absorption, polar surface area, and blood brain barrier penetration of some antiangiogenic agents, 2011 Structural Chem. 22 635-648.
S. Kováčová, L. Kováčiková, M. Lácová, A. Boháč, M. Sališová; Microwave assisted one pot synthesis of 7-substituted 2-(2-oxo-2H-chromen-3-yl)acetic acids as precursors of new anti-tumour compounds 2010 Chemical papers 64 806-811.
M. Lácová, R. Gašparová, P. Koiš, A. Boháč, H. M. El-Shaaer; A facile route to phenyl, phenylsulfanyl and phenylselanyl substituted pyrano[3,2-c]chromenes 2010 Tetrahedron 66 1410-1419.
L. Kováčiková, R. Gašparová, A. Boháč, M. Ďurana, M. Lácová; Synthesis of 3-phenyl-2H,5H-pyrano[3,2-c]chromen-2-one derivatives and their antineoplastic activit, 2010 ARKIVOC 11 188-203.
M. Pažický, V. Semak, B. Gášpár, A. Bílešová, M. Sališová, A. Boháč; Efficient synthesis of 2-alkyl-2-hydroxy-6-X-1-tetralones, 2008 ARKIVOC 8 225-241.
V. Semak, A. Boháč, M. Sališová, G. Addová, P. Danko; Preparation of alpha-methyl-gamma-butyrolactone: Mechanism of its formation and utilization in 2-methyl-1-tetralone synthesis, 2008 Chemical papers 62 275-280.
M. Lácová, H. Stankovičová, A. Boháč, B. Kotzianová, Convenient synthesis and unusual reactivity of 2-oxo-2H,5H-pyrano-[3,2-c]chromenes, 2008 Tetrahedron 64 9646-9653.
A. Almássy, Z. Benková, B. Horváth, A. Boháč; Fluxional behavior of methyl-substituted tricarbonyl(tropone)iron complexes and their different reactivity, 2008 J. of Organometal. Chem. 693 3223-3232.
M. Pažický, B. Gášpár, A. Solladié-Cavallo, M. Sališová, A. Boháč, M. Hutta, G. Addová; The epimerization of chiral alpha-amino acids by 2-hydroxy-2-methyl-1-tetralone template - The synthesis, conditions and mechanism of isomerization, 2006 Synthesis – Stuttgart 12 2013-2018.
V. Langer, D. Gyepesová, P. Mach, E. Scholtzová, B. Gášpár, M. Sališová, A. Boháč; Anti-2-Hydroxy-2-methyl-1-tetralone oxime: X-ray and density functional theory study, 2006 Acta Crystallographica Section C-Crystal Structure Communications 62 o199-o202, URL: http://www.blackwell-synergy.com/doi/full/10.1107/S0108270106005178.
O. Šedý, A. Solladié-Cavallo, M. Sališová, A. Boháč; Organické fosfazénové bázy a ich využitie v organickej chémii 2004 Chemické listy 98 964-969.
L. Soják, G. Addová, R. Kubinec, A. Kraus, A. Boháč; Capillary gas chromatography-mass spectrometry of all 93 acyclic octenes and their identification in fluid catalytic cracked gasoline, 2004 J. of Chromatography A 1025 237-253.
A. Almássy, B. Horváth, A. Boháč, M. Sališová, G. Addová, M. Rosenblum; Thermodynamic and kinetic parameters associated with the fluxional behavior of 2-methyl- and 2,6-dimethyltroponeiron tricarbonylcomplexes 2004 J. of Organometal. Chem. 689 1131-1138.
B. Horváth, A. Boháč, M. Sališová, E. Solčániová, M. Rosenblum; The synthesis of a hydroazulen-2-one skeleton-determination of the diastereoselectivity of [3+2] cycloaddition oftricarbonyl[4,5,6,7-n)-2-methyltropone]iron with E/Z-hapto1-(crotyl)Fp, 2002 J. of Organometal. Chem. 659 43-49.
A. Almássy, M. Pažický, A. Boháč, M. Sališová, G. Addová, M. Rosenblum; Synthesis of 2,6-Dimethyltropone - A New, Convenient Methodology from 2,6-Dimethylcyclohexanone 2002 Synthesis – Stuttgart 12 1695-1700.
G. Addová, L. Soják, R. Kubinec, A. Boháč; The comparison of GC behaviour of acyclic C5-C8 alkenes on squalane and silicone OV-1 stationary phases, 2000 Chemické listy 94 962-963.
R. Kubinec, L. Soják, R. Górová, G. Addová, A. Boháč; The separation of stereoisomers using recycle capillary gas chromatography, 1999 Enantiomer 4 345-350.
A. Boháč, M. Lettrichová, P. Hrnčiar, M. Hutta, Milan; Cyclic beta-diketones - precursors of chiral 1,3-cyclohexadiene iron carbonyl complexes. Ligard exchange - (-)-PPh2(O-HC(Me)-COOEt) - new, chiral, diastereomer-separating ligand in organoiron chemistry, 1996 J. of Organometal. Chem. 507 23-29.
A. Boháč; A transferable gas container (TGC) - improving the balloon inert atmosphere technique, 1995 J. of Chem. Education 72 1995 263-263.
Y. N. Ito, X. Ariza, A. K. Beck, A. Boháč, C. Ganter, R. E. Gawley, F. N. M. Kuhnle, J. Tuleja, Y. M. Wang, D. Seebach; Preparation and structural analysis of several newalpha,alpha,alpha,alpha-tetraaryl-1,3-dioxolane-4,5-dimethanols (TADDOL's) and TADDOL analogs, their evaluation as titanium liganst in the enantioselective addition of methyltitanium and diethylzinc reagents to benzaldehyde, and refinement of the mechanistic hypothesis, 1994 Helvetica Chimica Acta 77 2071-2110.
G. J. Matare, A. Boháč, P. Hrnčiar; Acetylation of beta-Diketone Copper(II) Chelates by Treatment with Ketene 1994 Synthesis – Stuttgart 4 381-382.
A. Boháč, P. Hrnčiar, J. Šraga; Acetylation of 2-acyl-1,3-indandiones with ketene and Determination of the Structure of Products 1992 Chemical papers 46 31-34.
A. Boháč, P. Hrnčiar; 3-Acetoxy-2-cykloalken-1-one by acetylation of cyclic 1,3-diketones with ketene; 1991 Collection of Czechoslovak Chem. Commun. 56 2879-2883.
A. Boháč, P. Hrnčiar; A Convenient Synthesis of 1,3-Diacetoxy-1,3-cycloalkadienes from Cyclic beta-diketones, 1991 Synthesis - Stuttgart. 10 881-882.
A. Boháč, A. Perjéssy, D. Loos, P. Hrnčiar; Elucidation of Acetylation Products of 2-Acyl-1,3-indanediones by Correlation of Infrared Spectral Data, 1991 Monatshefte für Chemie 122 943-948.
------------------------------------------------------------------------------
10th Joint Meeting on Medicinal Chemistry, 25-28 June 2017 Dubrovnik, Croatia venue: Hotel Sheraton in Srebreno (6 km from Dubrovnik) FLYER
http://www.jmmc2017.hr/
Lecturer: Andrej Boháč, Juraj Dobiaš, Peter Šramel, Miroslav Murár, Matúš Hlaváč (Comenius University, Biomagi, Bratislava, Slovakia)
Plenary lecture: From exceptional kinase conformation to dual kinase inhibitors – chemistry of 2-aminooxazoles